Corneal blindness and corneal damage are major global health problems affecting millions worldwide. Traditional treatment — corneal transplantation — is limited by the availability of donor tissue and surgical complexity. Enter Pandorum Technologies, an Indian biotech startup working on a groundbreaking regenerative solution called ‘Liquid Cornea’ that could transform how corneal damage is treated and how vision is restored.
This article provides an in‑depth, professional insight into the science, promise, and future of this innovation.
Understanding the Global Burden of Corneal Blindness
The cornea is the eye’s transparent outer covering that focuses light onto the retina. Damage to this tissue — from injury, infection, or degenerative disease — can cause blurred vision, severe discomfort, or complete blindness.
Globally, millions suffer from corneal opacity and related blindness. Estimates suggest that over 12 million people are waiting for corneal transplants, yet donor tissue is available for only a fraction of cases. This massive mismatch between demand and supply contributes to a persistent global health challenge.
Corneal blindness disproportionately affects regions with limited access to eye care infrastructure, including many developing countries where donor corneas are scarce. Millions more live with unilateral (one-eye) or bilateral (both eyes) corneal blindness.
What Is ‘Liquid Cornea’? A New Paradigm in Corneal Regeneration
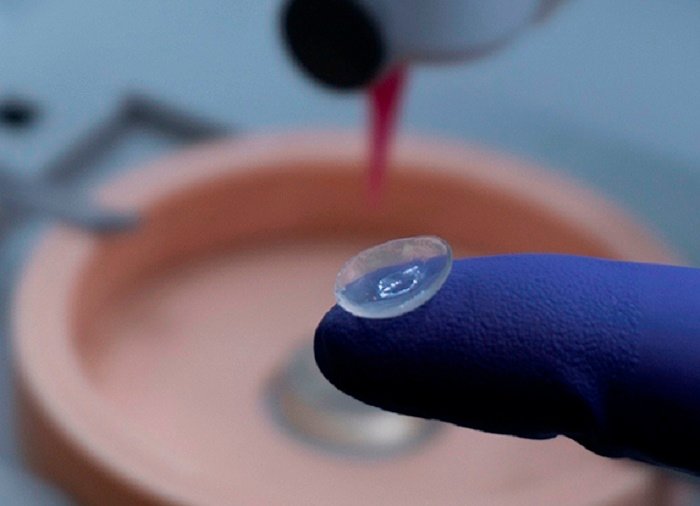
The ‘Liquid Cornea’ refers to a regenerative biomaterial therapy aiming to repair and regenerate damaged corneal tissue without the need for full corneal transplant surgery.
Unlike donor transplants, where whole corneal tissue from a deceased donor is surgically implanted, the Liquid Cornea is designed to be applied directly to the damaged cornea, where it integrates, promotes healing, and encourages scarless tissue regeneration.
This approach uses a biopolymeric solution and exosomes — tiny natural cell-derived vesicles that guide tissue repair and regeneration. When applied as drops, this solution solidifies and creates an environment conducive to healing, potentially leading to restoration of transparent, functioning corneal tissue over time.
The Science Behind the Innovation
Pandorum’s approach reflects advances in tissue engineering and regenerative medicine — fields that seek to mimic or restore living tissues through engineered solutions.
The core technology platform includes:
-
Bio‑mimetic materials that closely resemble the natural extracellular matrix of human tissues, providing mechanical support and chemical cues for cellular healing.
-
Exosome‑based regenerative agents, which are cellular messengers that instruct nearby cells to repair and regenerate tissue.
-
Advanced bio‑fabrication techniques, including 3D printing and hierarchical assembly, to create tissue-like structures.
In the case of Liquid Cornea, the therapeutic combines all these elements in a light‑activated hydrogel that can solidify on the eye within minutes of application and begin supporting tissue repair.
Pandorum Technologies: From Bangalore to Global Biotech
Founded by Dr. Tuhin Bhowmick and Arun Chandru, Pandorum Technologies is a Bangalore-based biotech company focused on tissue engineering and regenerative medicine.
The company started with a broad mission to develop functional human tissues through engineered biomaterials and advanced bio‑fabrication. Over time, its work has evolved to include clinical‑relevant therapeutic solutions — especially for corneal blindness.
Pandorum’s flagship program, called Kuragenx, embodies the Liquid Cornea concept. It has been developed in collaboration with leading clinicians and researchers from both India and the United States.
Funding and Development Milestones
Pandorum’s progress has been supported by several funding rounds and partnerships. In March 2024, the company secured USD 11 million in investment to advance Kuragenx toward clinical development.
In early 2026, the company raised USD 18 million in a Series B round to expand work on the Liquid Cornea technology and prepare for advanced stages of testing, including regulatory approvals and clinical trials.
These funds are supporting critical next steps such as manufacturing partnerships, regulatory submissions, and preparation for human clinical trials.
Clinical and Regulatory Pathway
While early testing of the Liquid Cornea has shown promising results in pre-clinical studies, including animal models, several layers of clinical validation and regulatory review remain.
Pandorum’s Kuragenx had received an Orphan Drug Designation from the U.S. Food and Drug Administration (FDA) for treating specific corneal conditions, signaling early regulatory recognition of its potential.
The company is currently moving toward first-in-human clinical trials, which are expected to occur after necessary safety and manufacturing evaluations are completed.
These steps are critical — success in human clinical trials and eventual regulatory approvals will determine whether Liquid Cornea can be deployed in hospitals and clinics worldwide.
Why This Matters: Potential Impact on Global Health
If successful, Liquid Cornea could transform corneal care in several key ways:
1. Reduced Dependence on Donor Tissue
By regenerating tissue rather than replacing it, Liquid Cornea could drastically cut reliance on donor corneas — easing pressure on eye banks and surgical systems with limited tissue availability.
2. Minimally Invasive Application
Unlike full surgical transplants, the therapy may be applied through a simple clinical procedure, potentially reducing risk, cost, and procedural complexity.
3. Increased Accessibility
Affordable and less surgical care could extend treatment to patients in low-resource settings where donor cornea shortages are greatest.
4. Faster Path to Vision Restoration
By promoting regenerative healing, this approach aims for functional vision recovery, not just structural repair.
Challenges and Future Outlook
Despite the promise, Liquid Cornea remains under development, and key hurdles lie ahead:
-
Clinical proof in humans — safety and efficacy must be confirmed in carefully controlled trials.
-
Regulatory approvals — approval pathways vary by country and require extensive evidence.
-
Manufacturing scale-up — producing consistent, high-quality bio-therapeutics for global distribution is complex.
Nevertheless, the rapid progress of Pandorum Technologies reflects a broader global trend toward regenerative and programmable medicine, where biological solutions are designed to heal and restore rather than simply replace.
Conclusion: A Vision for the Future
Pandorum’s Liquid Cornea represents a bold intersection of biotechnology, materials science, and regenerative medicine. If fully realized, this innovation could not only reshape the treatment of corneal blindness but also serve as a model for future regenerative therapies across medical fields.
With continued scientific validation and clinical development, India’s homegrown solution might one day help millions see clearly again — reducing blindness, expanding access to care, and redefining what is possible in ophthalmology.
totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com totosecom.com